Written by Daniel Mannina
December 15, 2020

Pride Month has been a celebration and showcase for underrepresented people and their allies to peacefully push for fair treatment and equality for 20 years. Established in 2000 to commemorate the 1969 New York City Stonewall Riots, this anniversary is particularly important amidst the current outcry for systemic civil rights reform.
Post-Stonewall, the rainbow pride flag was created as a symbol of solidarity for often repressed voices. Created by artist Gilbert Baker at the urging of Harvey Milk, one of the first openly gay officials elected in the U.S., the flag leverages a powerful natural phenomenon of hope. The pride flag’s six colors represent:
- Life (Red)
- Healing (Orange)
- Sunshine (Yellow)
- Nature (Green)
- Harmony (Blue)
- Spirit (Purple)
Understanding history and science are key to building a better tomorrow. Finding small ways to share both as a family creates opportunities for open conversations, collaborations and fun.
KITCHEN SCIENCE
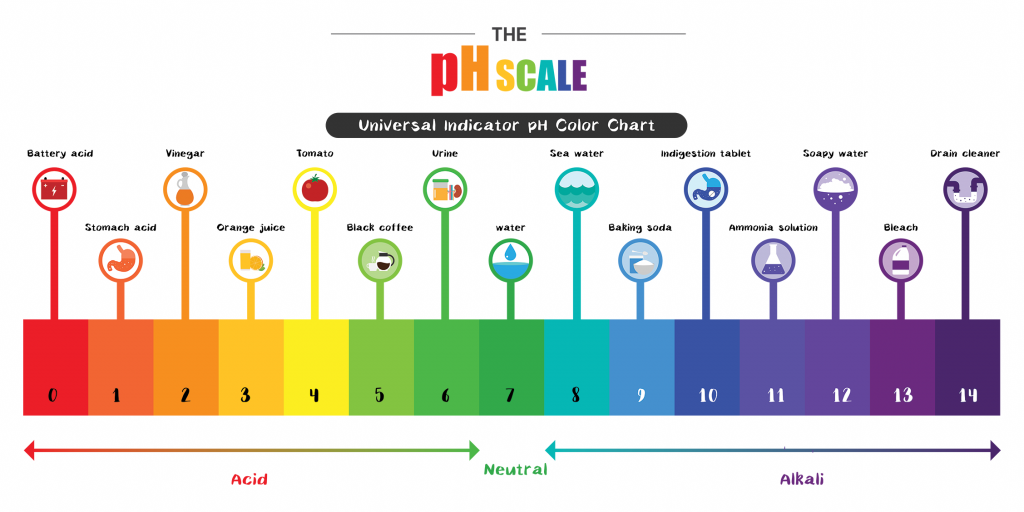
Create a river of cascading colors in this twist on the classic erupting volcano. This activity focuses on the pH scale, which measures hydrogen ions in a solution and allows you to determine if the solution is acidic, basic or neutral.
BASICS
This experiment requires 12 clear plastic cups, a two-quart container of white distilled vinegar, a 1-pound box of baking soda, one assorted pack of food coloring (containing red, blue, green and yellow primary colors), a tablespoon and a measuring cup.
- Place six plastic cups in a row; add two tablespoons of baking soda in each.
- Place another six plastic cups in front of the first set of cups which contain the baking soda. From left to right, put the following amounts of food coloring in each:
- Cup 1 (Red) – Five drops of red food coloring
- Cup 2 (Orange) – Three drops of yellow and two drops of red food coloring
- Cup 3 (Yellow) – Five drops of yellow food coloring
- Cup 4 (Green) – Five drops of green food coloring
- Cup 5 (Blue) – Five drops of blue food coloring
- Cup 6 (Purple) – One drop of blue and one drop of red food coloring
- Measure and pour a 1/2 cup of white distilled vinegar into each of the six cups which have the food coloring.
- One by one, pour the baking soda into each cup of colored vinegar until the acid-base reaction starts erupting like a volcano.
- BEFORE
- AFTER
WHAT IS HAPPENING?
In this experiment, we’re mixing vinegar, an acidic solution, with baking soda, which is a basic. Vinegar is low on the pH scale, which causes a big reaction when it meets baking soda which is high on the pH scale. The hydrogen atoms in the vinegar (acetic acid) rapidly replace the sodium atoms in the baking soda (sodium bicarbonate). The eruption is caused by the speed and bioenergetics – how cells transform energy – of the acid-base reaction.
Beyond creating a beautiful rainbow, the food coloring simulates the full spectrum of the pH scale. To actually test the pH of foods and household chemicals in a lab setting, items are first placed in a glass beaker. Separately, a special chemical known as a ‘universal indicator’ is mixed with distilled water.
Once the mix is poured into each glass beaker with food and household chemicals, the solution will turn into a color found on pH scale, showing how acidic, neutral or basic/alkali each item is.
Based on this information, look at the below pH scale and identify the two substances that were in this activity (white distilled vinegar and baking powder). What is their pH number/color?
SCALE-UP
Continue learning and growing during Pride Month by joining the Center for Humanities in an Urban Environment for a conversation on “Belonging: Reflections on Identity, Exile and Home” with inaugural poet and FIU alumnus Richard Blanco. The event will be held from 2 p.m. to 3:30 p.m. on June 24 via Zoom video conference.
Share your successes, failures and adjustments along with the final product with @FIUCASE on social media.
Central to every house there, is no better STEM classroom than the kitchen. From chemistry to biology to physics and beyond investigate the art and science behind culinary creations and everyday life. Follow FIU@Home on CASE News for more kitchen science.